Marsico Hall Microscopy Fellowship (MHMF.ORG)
|
|
Marsico Hall Microscopy Fellowship (MHMF.ORG) |
|
Olympus VS200 - Whole Slide Scanner with Slide Loader |
|
Location: Marsico Hall |
The VS200 ASW software was upgraded to version 3.4.1 on May 19, 2023 The free viewer for acquired data is OlyVIA (Windows only). Use version 4.2.1, 4.2, 4.1 or 4.1.1 (Windows 11) or OlyVIA 3.4.1 (Does not install on Windows 11). Older versions of OlyVIA will not open data acquired on the VS200 v3.4.1 or latre.Obtaining the OlyVIA viewer installation files:
Notices:
VS200 booking Calendar for trained users is here: |


30 Aug 2022 setup
Current setup with robotic slide feeder
on the right
VS200 booking Calendar is
here:
![]()
Points to note:
| Tray Number |
Tray serial number |
Height at slide position (um) | z offset (um) |
z offset (old) |
Status | ||||||
| 1 | 2 | 3 | 4 | 5 | 6 | avg | |||||
| 1 | 35001739 | 151 | 160 | 157 | 149 | 144 | 144 | 151 | 0 | 0 | In service |
| 2v4 | 35023858 | -14 | -6 | -10 | -6 | -1 | 11 | -4 | -155 | In service | |
| 3 | 35004731 | -81 | -69 | -57 | -35 | -15 | 6 | -42 | -193 | -225 | In service |
| 4 | 35004733 | -52 | -65 | -64 | -51 | -32 | 7 | -43 | -194 | -166 | In service |
| 5 | 35005150 | -64 | -51 | -36 | -1 | 21 | 45 | -14 | -165 | -137 | In service |
| 6 | 35005151 | 16 | 22 | 26 | 33 | 47 | 59 | 34 | -117 | -132 | In service |
| 7 | 35003711 | 17 | 34 | 40 | 45 | 48 | 51 | 39 | -112 | -119 | In service |
| 8 | 35004493 | 22 | 26 | 41 | 48 | 51 | 48 | 39 | -112 | -126 | In service |
| Tray Number |
Tray serial number |
Height at slide position (um)** | z offset* (um) |
z offset (old) |
Status | ||||||
| 1 | 2 | 3 | 4 | 5 | 6 | avg | |||||
| 1 | 35001739 | 172 | 156 | 156 | 152 | 169 | 169 | 161 | 0 | -1 | In service |
| 2 |
35001522 |
-35 | 20 | Recalled | |||||||
| 3 | 35004731 | -55 | -46 | -29 | -6 | 15 | 38 | -14 | -225 | -174 | In service |
| 4 | 35004733 | -28 | -45 | -45 | -39 | -16 | 25 | -25 | -166 | -139 | In service |
| 5 | 35005150 | -46 | -31 | -13 | 23 | 46 | 65 | 7 | -137 | -100 | In service |
| 6 | 35005151 | 40 | 41 | 47 | 55 | 69 | 81 | 56 | -132 | -87 | In service |
| 7 | 35003711 | 42 | 58 | 62 | 64 | 68 | 69 | 61 | -119 | In service | |
| 8 | 35004493 | 48 | 54 | 64 | 72 | 74 | 79 | 65 | -126 | In service | |
Mapping Drive Letters to servers (connecting to a serer, e.g. \\Minsky or CF-shared or CF-groups):
Important Considerations:
 If you remove
a slide tray with any slides with immersion oil present (60x scans)
belonging to the previous user or to yourself please:
If you remove
a slide tray with any slides with immersion oil present (60x scans)
belonging to the previous user or to yourself please:
 Hardening
mounting media needs to be well cured. No slides with wet media near lenses.
Clean off excess wet mounting by blotting with a tissue or cutting off
hardened material with
a razor blade. Slides with mounting media above the level of the cover
slip must not be be scanned.
Failure to do this will result in the scanner being unavailable for cleaning
and recalibration.
Hardening
mounting media needs to be well cured. No slides with wet media near lenses.
Clean off excess wet mounting by blotting with a tissue or cutting off
hardened material with
a razor blade. Slides with mounting media above the level of the cover
slip must not be be scanned.
Failure to do this will result in the scanner being unavailable for cleaning
and recalibration. General Considerations:
COVID-19 Mitigations:
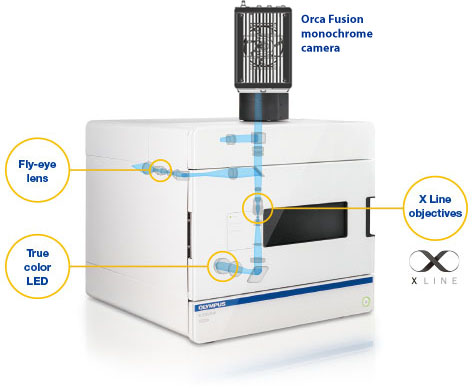
 The System:
The System:
| Position | Channel | Ex (nm) | BS (nm) | Em (nm) | Cube | Similar fluorophores | |
| 1 | transmitted | none | |||||
| 2 | DAPI | 392/23 (380-404) | 409 | 447/50 (422-472) | LED-DAPI-A-OMF | Hoechst | |
| 3 | FITC | 474/27 (460-488) | 495 | 525/45 (502-548) | LED-FITC-A-OMF-ZERO | GFP, Alexa 488, CY2, Opal 520 | |
| 4 | TRITC | 554/23 (542-566) | 573 | 609/54 (582-636) | LED-TRITC-A-OMF-ZERO | Opal 570, CY3, Alexa 555 | |
| 5 | mCherry | 578/21 (567-589) | 596 | 641/75 (604-679) | LED-mCherry-A-000-ZERO | Alexa 594, Opal 620, Texas Red | |
| 6 | CY5 | 635/18 (626-644) | 652 | 680/42 (659-701) | LED-CY5-A-OMF-ZERO | Alexa 633/647, Opal 650, Draq5 | |
| 7 | CY7 | 735/28 (721-749) | 757 | 809/81 (706-850) | LED-Cy7-A-000 | CY7Alexa 750, Opal 750 | |
| 8 | spare | none |
Objectives Mag. NA type WD corrections cover slip Immersion Pixel size
monochrome/Orca-FusionPixel size
color camera2x 0.06 PLN * 5.8 mm - #1.5 dry 3.25 um 2.738 um 4x 0.16 UPLXAPO 13 mm - #1.5 dry 1.625 um 1.369 um 10x 0.40 UPLXAPO 3.1 mm - #1.5 dry 0.650 um 0.5476 um 20x 0.80 UPLXAPO 0.60 mm - #1.5 dry 0.325 um 0.2738 um 40x 0.95 UPLXAPO 0.18 mm coverslip thickness 0.11-0.21 mm dry 0.1625 um 0.1369 um 60x 1.42 UPLXAPO O 0.15 mm - #1.5
oil 1 0.1083 um 0.09127 um 1 Standard immersion (carbon) oil (RI=1.515)
* Not suitable for CY5 or CY7
Viewing Software:
Using the system:
Network connections:
|
|
Last Updated: 2026-04-07 |